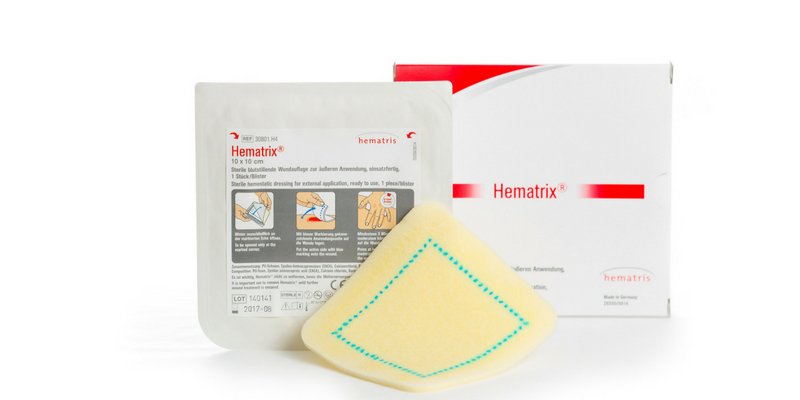
Hematrix® Active Patch
Hematrix® is an absorbent dressing containing substances
promoting blood clotting
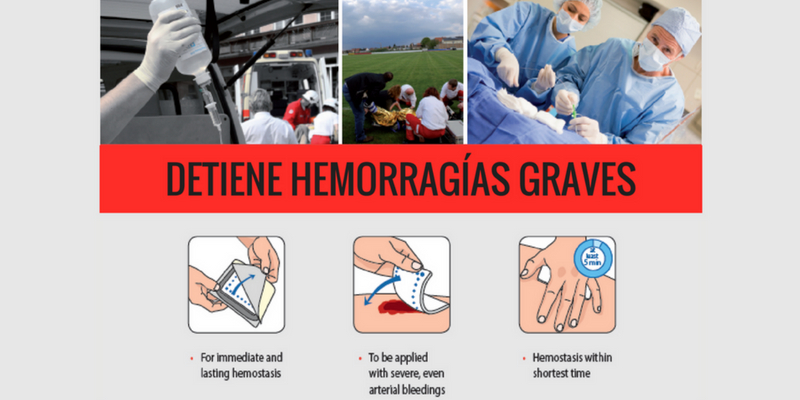
Is used on external wounds to stop even severe arterial or venous bleeding immediately
Prevents secondary bleedings
Allows earlier mobilisation of the patient
What can Hematrix® do?

The Hematrix® Active-Patch stops moderate to severe arterial and venous
bleeding:
• In hospitals and medical practices
• After all types of accident
• In the aftermath of catastrophes

Bleeding is usually treated using pressure dressings whose use calls for a high level of experience and absolute commitment, even in skilled personnel. The disadvantages of pressure dressings are the high cost in terms of time and the longer immobilisation of the patient. This is particularly important for patients with circulatory disorders as they are exposed to a greater risk when pressure is applied over a longer period

The Hematrix® Active-Patch stops arterial and venous bleeding within a few minutes. Longer-lasting compression is not necessary.
Rapid blood clotting means that the wound can be treated immediately.
Doubts?
We solve them here
↓
Composition Hematrix®
Hematrix® Active-Patch consists of a polyurethane sponge matrix impregnated with the epsilon-aminocaproic acid (EACA)) as an antifibrinolytic agent and has been widely used in medicine for many years, thrombin, a plasma protein and calcium chloride is an important co-factor for several clotting factors and supports the coagulation of the blood coming from the wound.
These constituents are held in the vesicles of the matrix (see diagram below), on the sponge’s internal surface. This means they are able to act rapidly when blood flows into the dressing.

Hematrix® Components
Polyurethane sponge
The basic structure of the Hematrix® Active-Patch is made from absorbent polyurethane which is impregnated with substances promoting blood clotting. This spongy material has been in use for many years in a wide variety of medical applications.
Epsilon-aminocaproic acid (EACA)
Epsilon-aminocaproic acid is well known as an antifibrinolytic agent and has been widely used in medicine for many years.
EACA also increases the activity of thrombin. We have demonstrated this effect in our own studies, upon which the product patent is based. The effect depends on the alkalising action of EACA. The activation of thrombin requires a pH of between 7.0 and 8.0. In a bleeding wound, however, the environment is slightly acidic. EACA is an ampholyte and therefore takes up protons, thus exerting a mild alkalising effect and supporting the activation of thrombin. The result is rapid and lasting clotting of the blood leaving the wound.
Calcium chloride (CaCl2)
Calcium chloride is an important co-factor for several clotting factors and supports the coagulation of the blood coming from the wound.
Thrombin
To speed up initial clotting the Hematrix® Active-Patch contains thrombin as a “starter”. This strengthens the clotting activity of the body’s own thrombin, which is present in the blood flowing out of the wound. Thrombin is a plasma protein. It acts as an enzyme, catalysing the conversion of fibrinogen into fibrin. This process represents the final phase in the coagulation cascade and leads to the formation of a blood clot.
The thrombin in the Hematrix® Active-Patch is of bovine origin. It has appropriate certification of quality and source, as required for human use.
How does Hematrix® work?
Because of its sponge-like structure the Hematrix® Active-Patch can soak up the blood that escapes after injury to a blood vessel. The many small vesicles making up the sponge provide a large surface area and numerous micro-areas for reactions to occur and thus exert a pseudo-catalytic effect on blood clotting.
This leads to an increase in the reactivity of the blood cell structures and associated release of coagulant factors. As a result, the
whole blood clotting process – the coagulation cascade – is activated. The thrombin in the Hematrix® Active-Patch intensifies the action of the body’s own prothrombin/ thrombin system and so accelerates blood clotting.
The activation of thrombin requires a pH of 7.0 – 8.0. The epsilon aminocaproic acid (EACA) in the Hematrix® Active-Patch supports the activation of thrombin – both that contained in the Patch and that produced by the body – by slightly alkalising the blood flowing into the spongy matrix. The fibrinogen in the blood leaving the wound is then converted into fibrin.
EACA possesses antifibrinolytic properties which have been known and applied in medicine for many years and which help to stabilise the blood clot. The ready-to-use dressing does not contain any fibrinogen of its own which could be converted into fibrin and stick it to the wound. Once bleeding has been stopped, the design of the Hematrix® Active-Patch and its combination of constituents make it possible to remove the dressing from the wound without causing secondary bleeding. The blood clot forms on the lesion in the blood vessel and remains there. This property is crucial for the final treatment of the wound once bleeding has been stopped.
The calcium chloride which is also present in the sponge acts as a co-factor supporting the blood clotting process (the coagulation cascade) and helps to stop bleeding.
Once started, blood clotting normally progresses in a retrograde fashion, working back towards the wound. The speed of this process depends on the intensity of the bleeding. The blood clotting process triggered by the Hematrix® Active-Patch works according to the same principle in the case of a deeper cut and/or puncture opening. The thrombin in the Hematrix® Active-Patch cannot move against the outward flow of blood to come into contact with the wound and cannot diffuse into the wound because of its large molecular size. It only supports the clotting of the blood that flows out of the wound. This reduces further bleeding into the surrounding tissue (bruising).
If traces of EACA diffuse out of the blood clot held in the sponge and reach the wound after bleeding had stopped, the amounts that could be involved are very small and do not present a risk to the patient.
Where does Hematrix® act?
Constituents of Hematrix® and the coagulation cascade:
(Schematic representation)

How is Hematrix® used?
This Hematrix® Active-Patch is indicated for the stoppage of bleeding from external wounds.
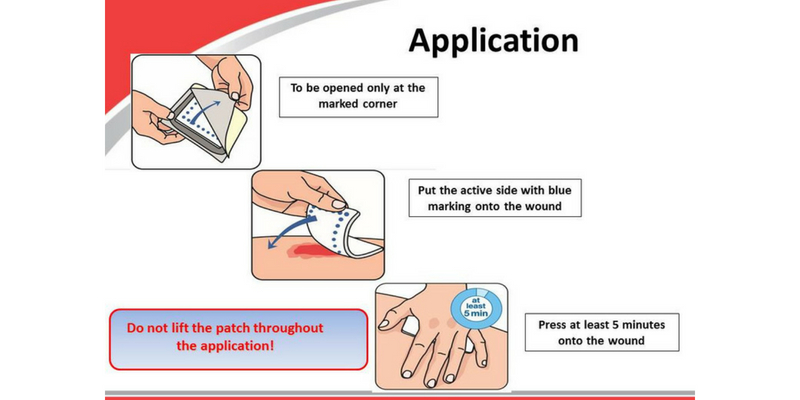
The Hematrix® Active-Patch is ready to use. It is positioned so that the side marked with a blue square (the active side of the dressing) is in contact with the wound.
After injury, or in cases of iatrogenically induced bleeding, the Hematrix® Active-Patch should be placed on the wound immediately. The blood flowing from the wound must come into contact with the Patch so that it can initiate satisfactory blood clotting.
The Hematrix® Active-Patch begins to act as soon as it has made contact with the blood. To make this happen the dressing must be pressed onto the wound and held firmly in place for a few minutes. This enables the blood to enter the spongy matrix. Blood clotting is initiated both within the matrix and on its surface, and progresses in a retrograde direction towards the lesion in the blood vessel. The spongy matrix also possesses a large capacity to absorb blood and wound secretions.
The Hematrix® Active-Patch can then be fixed on the wound with a bandage or adhesive tape. It should remain there until the wound is given further treatment.
The material making up the matrix of the Hematrix® Active-Patch is very flexible and nonabsorbable. It should therefore be used only on external injuries. Under no circumstances should it remain in the body. It is important that the dressing be pressed firmly onto the wound for at least 3-5 minutes, depending on the severity of the bleeding. Just placing the dressing on the wound and fixing it with adhesive tape or a bandage is not sufficient! This applies both for arterial and for venous bleeding.
How was Hematrix® tested?
1) Preclinical testing
The effect of the Hematrix® Active-Patch on bleeding was tested in pigs. The versions tested were the same in terms of composition and manufacturing quality as the dressings used on patients in clinical trials. A scalpel was used to create an experimental wound and a wide opening in the femoral artery. The massive bleeding that resulted was stopped completely in a few minutes by immediately pressing a Hematrix® Active-Patch onto the wound. No secondary bleeding occurred, even when the dressing was carefully separated from the wound. This is not normally done until the wound receives secondary treatment. However, it was not possible to stop the bleeding with a control dressing.
The direct comparison with the control dressing was impressive in demonstrating that the Hematrix® Active-Patch can quickly stop bleeding and thus reduce blood loss. Because of its large absorption capacity the polyurethane matrix is also able to take up wound secretions, improving wound hygiene and aiding the further treatment of the wound.
2) Clinical testing
Appropriate trials were carried out to determine the efficacy and safety of the Hematrix® Active-Patch when used to stop bleeding in humans. On the basis of the results of the animal experiments, the haemostatic effect was tested on arterial bleeding such as that following coronary angiography.
The catheter was inserted via the femoral artery. The removal of the catheter in this procedure causes arterial bleeding which needs to be stopped. Normally a pressure dressing is applied postoperatively for several hours.
Despite the fact that bleeding is successfully stopped, this is often associated with bruising and other problems for the patient (such as immobilisation and circulatory disturbance.
Note: The clinical functioning of the Hematrix® Active-Patch to stop bleeding after heart catheter use was tested as required by the authorisation/ certification institution and the ethics committee in relation to standardised haemostasis (wound treatment) in everyday clinical practice. The central point was that haemostasis was confirmed, not only in the arterial bleeding model but also for a non-superficial source of bleeding.
The study was carried out according to a testing protocol approved by the ethics committee. It involved 100 patients at a major cardiology centre. The haemostatic efficacy of a 10 x 10 cm dressing was tested.
The study’s end point was the cessation of bleeding after removal of a catheter from the femoral artery. For this purpose the Hematrix® Active-Patch was pressed firmly onto the site of bleeding for five minutes with the hand. For the dressing to exert its haemostatic effect, it must make contact with the blood flowing from the wound.
The protocol reports that the artery was not allowed to be clamped upstream additionally in the usual way to maintain blood circulation. A pressure dressing was then applied as an additional safeguard, but for two hours rather than for the six hours that were usual at this hospital. The patients spent this period in the observation room.
Results
In 93% of the patients, bleeding stopped completely after the five minute period for which a Hematrix® Active-Patch was pressed against the wound. Bleeding did not restart in the subsequent hours.
Only six patients (6%) required another Hematrix® Active-Patch. In five of these six cases this was because the first Hematrix® Active-Patch had not been applied correctly. In only one of the patients was it not possible to stop the bleeding. This patient had to be treated with a pressure dressing.
How safe is Hematrix®?
The Hematrix® Active-Patch is a class III medical product for external use to stop bleeding. A classification report on Hematrix® is available (Klassifizierungsgutachten zum Medizinprodukt Hematrix®) as required by the German law on medical products (Medizinproduktegesetz, MPG).
The Hematrix® Active-Patch is not inserted into the body. As a result it does not interact with the patient‘s metabolic processes. Clotting is promoted exclusively in the blood flowing from the wound. All the constituents of the Hematrix® Active-Patch have been in medical
use for many years. Their trouble-free record serves to underline the safety of this product.
Product form / design
The patient’s body only comes into contact with the surface of the Hematrix ® Active-Patch. The special manufacturing process ensures that the active substances occur mainly on the inner surface of the matrix vesicles and cannot move upstream against the flow of blood to reach the wound.
Constituents
After coagulation, which occurs immediately, the constituents of the Hematrix ® Active-Patch remain with the clotted blood in the product matrix.
Thrombin
Being a high molecular weight protein, thrombin does not diffuse out of the dressing because of its large molecular size but remains within the blood clot. It is also forced back by the blood flowing out of the wound.
Antigen sensitisation as a result of the bovine thrombin is therefore impossible. Bovine thrombin is used in a variety of wound treatment products currently on the market without causing problems, a fact emphasising the safety of this treatment. Depending on the application and the product, the thrombin may even be introduced into the body to treat the wound. Studies on a large number of patients confirm that intolerance does not occur, even when nonhuman thrombin enters the human body The quality and origins of the thrombin conform to all the pharmaceutical specifications covering its use in humans. The manufacturer has obtained a Certificate of Suitability from the European Directorate for the Quality of Medicines.
Calcium chloride (CaCl2)
Calcium chloride also occurs in the human body. It is an important co-factor in the blood clotting process.
Epsilon aminocaproic acid (EACA)
As an individual substance EACA has been in use for many years in medical applications, particularly in heart surgery. It is used as an antifibrinolytic to treat or prevent haemorrhaging and to inhibit fibrinolytic processes. The substance on its own is unproblematic, even when given intravenously at high doses of several gram.
Application
The Hematrix® Active-Patch is only authorised for external use. This is because it is made from polyurethane material which is biocompatible but not bioresorbable. As a result the Hematrix® Active-Patch cannot be left in the body after surgery.
When applying the Hematrix® Active-Patch the user should be aware that the patch must make direct contact with blood in order to be effective.
The blood needs to penetrate into the spongy matrix. The dressing must be pressed firmly against the bleeding wound for several minutes. Only then, after release of pressure, can the dressing be fixed over the wound with adhesive foil or tape, or with a bandage, until further treatment can be given.
The dressing is pressed onto the bleeding wound. The active side which is marked with a blue square (see picture, p. 9), should face
towards the wound. To stop bleeding immediately it must be used this way round.
The dressing should remain in position until the wound can be treated properly.
Who can benefit from Hematrix®?
The Hematrix® Active-Patch stops bleeding immediately in patients with unimpaired blood clotting. It can also be used effectively in patients on anticoagulant therapy.
For these patients it is especially important to stop bleeding at once because every bleed can mean massive loss of blood. Studies have shown that the Hematrix® Active-Patch can be used successfully with this patient group, yielding a particularly convincing response rate of 93%.
Compression dressings are usually contraindicated in patients with disorders of arterial blood flow. The Hematrix® Active-Patch makes it possible to treat even arterial bleeding without applying extreme compression over a long period.
What are the advantages of Hematrix®?
Medical advantages
- stops bleeding within 2-5 minutes
- also stops arterial bleeding
- no secondary bleeding
- does not stick to the wound
- reduced bruising
- no restriction of blood flow
- improved wound healing
- improved wound hygiene
- useful in cases of multiple injury
Economic advantages
- less time required to stop bleeding and treat the wound
- patient can be transported more easily
- earlier mobilisation of the patient
Product information
Composition per patch
-
Thrombin
-
CaCl2
-
Epsilon-aminocaproic acid (EACA)
Form
Wound dressing
Size
- 10cm x 10cm
- 5cm x 5cm
Materials
GALLERY
DOWNLOAD INFO